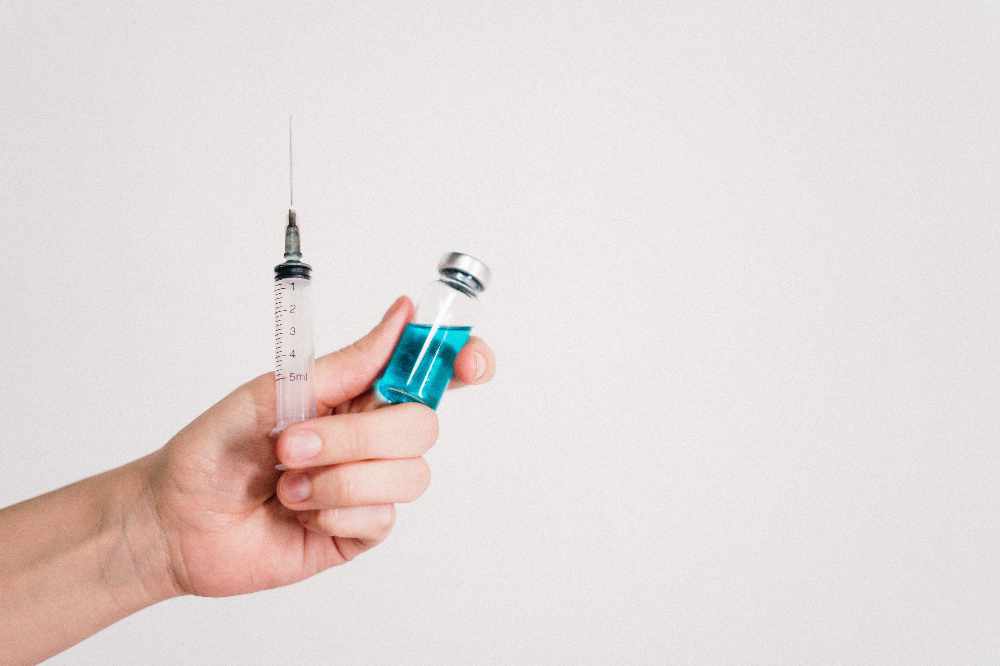
Moderna is seeking Health Canada authorization for COVID-19 vaccine for kids as young as six months old.
Health Canada is reviewing a request for a COVID-19 vaccine for young children.
Moderna has applied for authorization of a COVID-19 shot for children between the ages of six months and five years old.
Earlier today, Health Canada updated its website, confirming the request has been received.
This comes one day after the company filed a similar request to the U.S. Food and Drug Administration, and the same day the Canadian government announced a new Moderna facility coming to Montreal to produce mRNA vaccines.
Last month, the company's vaccine for kids aged 6 to 11 was approved.
At the time, Moderna said trials indicate a low-dose of the shot works in babies, toddlers and preschoolers.


 Moyer Farms Making $12M Investment
Moyer Farms Making $12M Investment
 Niagara Falls Going Green for St. Patrick's Day
Niagara Falls Going Green for St. Patrick's Day
 St. Catharines RIDE Campaign
St. Catharines RIDE Campaign
 Davies Challenges Gov't on EV Support
Davies Challenges Gov't on EV Support
 Welland's Plan for Hospital Contribution
Welland's Plan for Hospital Contribution
 New GO Station Coming to Niagara
New GO Station Coming to Niagara
 Most Serious Collisions in Summer: Region
Most Serious Collisions in Summer: Region
 Coach Service Added to Airport
Coach Service Added to Airport